Nápady Atom Mass And Charge
Nápady Atom Mass And Charge. Mass and charge of the electron for comparison purposes. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. One way that they differ is in terms of mass.
Nejchladnější Difference Between Atomic Mass And Mass Number
The charge to mass ratio (e/m) for. When we write the symbol for an atom, we can place its mass number at … However, the nucleus is made from collections of two smaller particles: The electrons are fundamental particles.Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons:
Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: One way that they differ is in terms of mass. The charge to mass ratio of an electron is denoted by the following formula : The charge to mass ratio (e/m) for. Has no overall charge, because it has an equal number of protons and electrons. The electrons are fundamental particles. However, the nucleus is made from collections of two smaller particles:

= 1.758820 × 1011 c/kg. E = magnitude of the charge of the electron in coulombs. Atomic structure the distribution of mass and charge within an atom: When we write the symbol for an atom, we can place its mass number at … Although similar in mass, protons are positively charged, while neutrons have no charge. The electrons are fundamental particles. Where in, m = mass of electron in kg. Atoms have a tiny central nucleus with electrons whizzing around outside it. Mass and charge of the electron for comparison purposes. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. Has no overall charge, because it has an equal number of protons and electrons.

Where in, m = mass of electron in kg. Has no overall charge, because it has an equal number of protons and electrons. The charge to mass ratio (e/m) for. = 1.758820 × 1011 c/kg. Charge by mass ratio of an electron.. Atomic structure the distribution of mass and charge within an atom:

Although similar in mass, protons are positively charged, while neutrons have no charge. The electrons are fundamental particles. Where in, m = mass of electron in kg. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Charge by mass ratio of an electron. The number of protons, neutrons and electrons in a. Mass and charge of the electron for comparison purposes. The charge to mass ratio (e/m) for. However, the nucleus is made from collections of two smaller particles:

Atomic structure the distribution of mass and charge within an atom: However, the nucleus is made from collections of two smaller particles: Although similar in mass, protons are positively charged, while neutrons have no charge.. Atomic structure the distribution of mass and charge within an atom:
%252C_black_and_white.png?revision=2)
Although similar in mass, protons are positively charged, while neutrons have no charge. Charge by mass ratio of an electron. When we write the symbol for an atom, we can place its mass number at … Where in, m = mass of electron in kg. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Atoms have a tiny central nucleus with electrons whizzing around outside it. One way that they differ is in terms of mass. The charge to mass ratio (e/m) for. However, the nucleus is made from collections of two smaller particles: E = magnitude of the charge of the electron in coulombs. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: = 1.758820 × 1011 c/kg.

One way that they differ is in terms of mass. Although similar in mass, protons are positively charged, while neutrons have no charge. When we write the symbol for an atom, we can place its mass number at … Atomic structure the distribution of mass and charge within an atom: E = magnitude of the charge of the electron in coulombs. The number of protons, neutrons and electrons in a. However, the nucleus is made from collections of two smaller particles: The electrons are fundamental particles. The charge to mass ratio of an electron is denoted by the following formula :. The charge to mass ratio of an electron is denoted by the following formula :

Atomic structure the distribution of mass and charge within an atom: Mass and charge of the electron for comparison purposes. When we write the symbol for an atom, we can place its mass number at … The number of protons, neutrons and electrons in a.

Scientists define this amount of mass as one atomic mass unit (amu) or one dalton.. Mass and charge of the electron for comparison purposes. However, the nucleus is made from collections of two smaller particles: They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. The charge to mass ratio (e/m) for.. The charge to mass ratio of an electron is denoted by the following formula :

Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. Although similar in mass, protons are positively charged, while neutrons have no charge. = 1.758820 × 1011 c/kg. The electrons are fundamental particles. One way that they differ is in terms of mass.. However, the nucleus is made from collections of two smaller particles:

The charge to mass ratio (e/m) for. The charge to mass ratio of an electron is denoted by the following formula :
.PNG)
However, the nucleus is made from collections of two smaller particles:.. Although similar in mass, protons are positively charged, while neutrons have no charge. The charge to mass ratio (e/m) for. Atoms have a tiny central nucleus with electrons whizzing around outside it. E = magnitude of the charge of the electron in coulombs. Has no overall charge, because it has an equal number of protons and electrons. Mass and charge of the electron for comparison purposes.. E = magnitude of the charge of the electron in coulombs.

The charge to mass ratio (e/m) for. E = magnitude of the charge of the electron in coulombs. However, the nucleus is made from collections of two smaller particles:

However, the nucleus is made from collections of two smaller particles:. The charge to mass ratio (e/m) for. However, the nucleus is made from collections of two smaller particles: Although similar in mass, protons are positively charged, while neutrons have no charge.

The electrons are fundamental particles. The charge to mass ratio of an electron is denoted by the following formula :. The electrons are fundamental particles.

They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.. The charge to mass ratio of an electron is denoted by the following formula : They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. The electrons are fundamental particles. Atomic structure the distribution of mass and charge within an atom:. One way that they differ is in terms of mass.

E = magnitude of the charge of the electron in coulombs. Although similar in mass, protons are positively charged, while neutrons have no charge. The electrons are fundamental particles. The charge to mass ratio of an electron is denoted by the following formula : Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Where in, m = mass of electron in kg.

The number of protons, neutrons and electrons in a. . The charge to mass ratio (e/m) for.

Has no overall charge, because it has an equal number of protons and electrons.. When we write the symbol for an atom, we can place its mass number at … Charge by mass ratio of an electron. Atoms have a tiny central nucleus with electrons whizzing around outside it. Mass and charge of the electron for comparison purposes. Has no overall charge, because it has an equal number of protons and electrons. Although similar in mass, protons are positively charged, while neutrons have no charge.

When we write the symbol for an atom, we can place its mass number at ….. Mass and charge of the electron for comparison purposes. One way that they differ is in terms of mass. Charge by mass ratio of an electron. When we write the symbol for an atom, we can place its mass number at … However, the nucleus is made from collections of two smaller particles: They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. = 1.758820 × 1011 c/kg. However, the nucleus is made from collections of two smaller particles:
They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. The charge to mass ratio of an electron is denoted by the following formula : = 1.758820 × 1011 c/kg. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons:

The electrons are fundamental particles.. .. When we write the symbol for an atom, we can place its mass number at …

Has no overall charge, because it has an equal number of protons and electrons... The electrons are fundamental particles.. Mass and charge of the electron for comparison purposes.

= 1.758820 × 1011 c/kg. The number of protons, neutrons and electrons in a. Has no overall charge, because it has an equal number of protons and electrons. One way that they differ is in terms of mass.

Atomic structure the distribution of mass and charge within an atom:. The charge to mass ratio (e/m) for. = 1.758820 × 1011 c/kg. The charge to mass ratio of an electron is denoted by the following formula : Although similar in mass, protons are positively charged, while neutrons have no charge. Although similar in mass, protons are positively charged, while neutrons have no charge.

When we write the symbol for an atom, we can place its mass number at …. Although similar in mass, protons are positively charged, while neutrons have no charge. When we write the symbol for an atom, we can place its mass number at … However, the nucleus is made from collections of two smaller particles: The number of protons, neutrons and electrons in a. Mass and charge of the electron for comparison purposes. The charge to mass ratio of an electron is denoted by the following formula : Atomic structure the distribution of mass and charge within an atom: They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Atoms have a tiny central nucleus with electrons whizzing around outside it. = 1.758820 × 1011 c/kg.. Where in, m = mass of electron in kg.

The charge to mass ratio of an electron is denoted by the following formula : The number of protons, neutrons and electrons in a. Where in, m = mass of electron in kg. One way that they differ is in terms of mass. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Although similar in mass, protons are positively charged, while neutrons have no charge. Atomic structure the distribution of mass and charge within an atom: Atoms have a tiny central nucleus with electrons whizzing around outside it... One way that they differ is in terms of mass.

Mass and charge of the electron for comparison purposes.. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.. Although similar in mass, protons are positively charged, while neutrons have no charge.

E = magnitude of the charge of the electron in coulombs. However, the nucleus is made from collections of two smaller particles: Mass and charge of the electron for comparison purposes. When we write the symbol for an atom, we can place its mass number at … The electrons are fundamental particles. The number of protons, neutrons and electrons in a. The charge to mass ratio (e/m) for. The charge to mass ratio of an electron is denoted by the following formula : Atoms have a tiny central nucleus with electrons whizzing around outside it. Charge by mass ratio of an electron... The electrons are fundamental particles.

Atomic structure the distribution of mass and charge within an atom:. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. However, the nucleus is made from collections of two smaller particles: The charge to mass ratio (e/m) for. Atoms have a tiny central nucleus with electrons whizzing around outside it. The number of protons, neutrons and electrons in a. Although similar in mass, protons are positively charged, while neutrons have no charge.

Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: E = magnitude of the charge of the electron in coulombs. Mass and charge of the electron for comparison purposes. The electrons are fundamental particles. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. One way that they differ is in terms of mass.

The charge to mass ratio (e/m) for. The charge to mass ratio of an electron is denoted by the following formula : The charge to mass ratio (e/m) for. Charge by mass ratio of an electron. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton.

The electrons are fundamental particles. Mass and charge of the electron for comparison purposes. Has no overall charge, because it has an equal number of protons and electrons. Charge by mass ratio of an electron. Where in, m = mass of electron in kg. However, the nucleus is made from collections of two smaller particles: = 1.758820 × 1011 c/kg. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.. Although similar in mass, protons are positively charged, while neutrons have no charge.

One way that they differ is in terms of mass. . Charge by mass ratio of an electron.

Where in, m = mass of electron in kg.. The charge to mass ratio (e/m) for. = 1.758820 × 1011 c/kg. One way that they differ is in terms of mass. Charge by mass ratio of an electron. E = magnitude of the charge of the electron in coulombs. The charge to mass ratio of an electron is denoted by the following formula : Although similar in mass, protons are positively charged, while neutrons have no charge. The electrons are fundamental particles. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. One way that they differ is in terms of mass.

E = magnitude of the charge of the electron in coulombs.. One way that they differ is in terms of mass. Although similar in mass, protons are positively charged, while neutrons have no charge. The charge to mass ratio (e/m) for. Mass and charge of the electron for comparison purposes. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. The electrons are fundamental particles. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: However, the nucleus is made from collections of two smaller particles: Has no overall charge, because it has an equal number of protons and electrons. When we write the symbol for an atom, we can place its mass number at … They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.

When we write the symbol for an atom, we can place its mass number at … = 1.758820 × 1011 c/kg. However, the nucleus is made from collections of two smaller particles: The charge to mass ratio (e/m) for.. E = magnitude of the charge of the electron in coulombs.

E = magnitude of the charge of the electron in coulombs. The charge to mass ratio of an electron is denoted by the following formula :

However, the nucleus is made from collections of two smaller particles: Atoms have a tiny central nucleus with electrons whizzing around outside it.

The electrons are fundamental particles. Atomic structure the distribution of mass and charge within an atom: Has no overall charge, because it has an equal number of protons and electrons. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Atoms have a tiny central nucleus with electrons whizzing around outside it. The electrons are fundamental particles. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.

Where in, m = mass of electron in kg. The charge to mass ratio (e/m) for. Mass and charge of the electron for comparison purposes. Where in, m = mass of electron in kg. Although similar in mass, protons are positively charged, while neutrons have no charge.
Atomic structure the distribution of mass and charge within an atom:.. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. The number of protons, neutrons and electrons in a. When we write the symbol for an atom, we can place its mass number at … The electrons are fundamental particles. E = magnitude of the charge of the electron in coulombs. However, the nucleus is made from collections of two smaller particles:. Mass and charge of the electron for comparison purposes.
However, the nucleus is made from collections of two smaller particles: The electrons are fundamental particles. The charge to mass ratio of an electron is denoted by the following formula : Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: When we write the symbol for an atom, we can place its mass number at … However, the nucleus is made from collections of two smaller particles: Where in, m = mass of electron in kg. E = magnitude of the charge of the electron in coulombs. One way that they differ is in terms of mass. Has no overall charge, because it has an equal number of protons and electrons. Atomic structure the distribution of mass and charge within an atom: The number of protons, neutrons and electrons in a.

Scientists define this amount of mass as one atomic mass unit (amu) or one dalton... E = magnitude of the charge of the electron in coulombs.

Has no overall charge, because it has an equal number of protons and electrons... Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. Has no overall charge, because it has an equal number of protons and electrons. The number of protons, neutrons and electrons in a. Where in, m = mass of electron in kg. Atomic structure the distribution of mass and charge within an atom: The charge to mass ratio of an electron is denoted by the following formula : However, the nucleus is made from collections of two smaller particles:. Charge by mass ratio of an electron.

Has no overall charge, because it has an equal number of protons and electrons. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Charge by mass ratio of an electron. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Atomic structure the distribution of mass and charge within an atom: The electrons are fundamental particles... The number of protons, neutrons and electrons in a.

The charge to mass ratio of an electron is denoted by the following formula :. However, the nucleus is made from collections of two smaller particles: One way that they differ is in terms of mass. Atoms have a tiny central nucleus with electrons whizzing around outside it. Charge by mass ratio of an electron. E = magnitude of the charge of the electron in coulombs. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton.. Charge by mass ratio of an electron.

Atomic structure the distribution of mass and charge within an atom: Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. The electrons are fundamental particles. When we write the symbol for an atom, we can place its mass number at …. Charge by mass ratio of an electron.

Mass and charge of the electron for comparison purposes... Atomic structure the distribution of mass and charge within an atom: Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. However, the nucleus is made from collections of two smaller particles: Mass and charge of the electron for comparison purposes. = 1.758820 × 1011 c/kg.. However, the nucleus is made from collections of two smaller particles:

E = magnitude of the charge of the electron in coulombs. Mass and charge of the electron for comparison purposes. The charge to mass ratio (e/m) for. Atomic structure the distribution of mass and charge within an atom: The electrons are fundamental particles. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: One way that they differ is in terms of mass. Has no overall charge, because it has an equal number of protons and electrons. Atoms have a tiny central nucleus with electrons whizzing around outside it. The charge to mass ratio of an electron is denoted by the following formula : They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.. Atomic structure the distribution of mass and charge within an atom:
= 1.758820 × 1011 c/kg. Mass and charge of the electron for comparison purposes. One way that they differ is in terms of mass. E = magnitude of the charge of the electron in coulombs. The charge to mass ratio (e/m) for. The charge to mass ratio of an electron is denoted by the following formula : The electrons are fundamental particles.. The electrons are fundamental particles.
.PNG)
Scientists define this amount of mass as one atomic mass unit (amu) or one dalton... The number of protons, neutrons and electrons in a. However, the nucleus is made from collections of two smaller particles: Although similar in mass, protons are positively charged, while neutrons have no charge. Charge by mass ratio of an electron. Mass and charge of the electron for comparison purposes. = 1.758820 × 1011 c/kg. Although similar in mass, protons are positively charged, while neutrons have no charge.

Charge by mass ratio of an electron. Atomic structure the distribution of mass and charge within an atom: They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Has no overall charge, because it has an equal number of protons and electrons. One way that they differ is in terms of mass. E = magnitude of the charge of the electron in coulombs. The charge to mass ratio (e/m) for. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons:.. One way that they differ is in terms of mass.

However, the nucleus is made from collections of two smaller particles:. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: E = magnitude of the charge of the electron in coulombs. The charge to mass ratio (e/m) for. Charge by mass ratio of an electron. Where in, m = mass of electron in kg.. Atoms have a tiny central nucleus with electrons whizzing around outside it.

Atomic structure the distribution of mass and charge within an atom: . Scientists define this amount of mass as one atomic mass unit (amu) or one dalton.

Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. The electrons are fundamental particles. Charge by mass ratio of an electron. The number of protons, neutrons and electrons in a. The charge to mass ratio (e/m) for. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.. Atomic structure the distribution of mass and charge within an atom:

Although similar in mass, protons are positively charged, while neutrons have no charge.. = 1.758820 × 1011 c/kg. Atoms have a tiny central nucleus with electrons whizzing around outside it. Where in, m = mass of electron in kg. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. E = magnitude of the charge of the electron in coulombs. When we write the symbol for an atom, we can place its mass number at … The charge to mass ratio (e/m) for. However, the nucleus is made from collections of two smaller particles:.. Mass and charge of the electron for comparison purposes.

The charge to mass ratio of an electron is denoted by the following formula : E = magnitude of the charge of the electron in coulombs. However, the nucleus is made from collections of two smaller particles: Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Although similar in mass, protons are positively charged, while neutrons have no charge. The charge to mass ratio of an electron is denoted by the following formula : The charge to mass ratio (e/m) for. Atoms have a tiny central nucleus with electrons whizzing around outside it. Where in, m = mass of electron in kg. Mass and charge of the electron for comparison purposes.

The number of protons, neutrons and electrons in a. Where in, m = mass of electron in kg. When we write the symbol for an atom, we can place its mass number at … The charge to mass ratio (e/m) for. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Mass and charge of the electron for comparison purposes. One way that they differ is in terms of mass.. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.

The charge to mass ratio of an electron is denoted by the following formula : The charge to mass ratio of an electron is denoted by the following formula : When we write the symbol for an atom, we can place its mass number at … The charge to mass ratio (e/m) for. However, the nucleus is made from collections of two smaller particles: Where in, m = mass of electron in kg. Mass and charge of the electron for comparison purposes. Has no overall charge, because it has an equal number of protons and electrons. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: One way that they differ is in terms of mass. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. The charge to mass ratio of an electron is denoted by the following formula :

One way that they differ is in terms of mass. The charge to mass ratio (e/m) for. The electrons are fundamental particles. However, the nucleus is made from collections of two smaller particles: When we write the symbol for an atom, we can place its mass number at … Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: E = magnitude of the charge of the electron in coulombs. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Although similar in mass, protons are positively charged, while neutrons have no charge. Atoms have a tiny central nucleus with electrons whizzing around outside it.

Scientists define this amount of mass as one atomic mass unit (amu) or one dalton... One way that they differ is in terms of mass. The electrons are fundamental particles. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Atomic structure the distribution of mass and charge within an atom: When we write the symbol for an atom, we can place its mass number at …. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton.

Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons:. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. The number of protons, neutrons and electrons in a. The charge to mass ratio (e/m) for. Charge by mass ratio of an electron. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.

The charge to mass ratio (e/m) for. = 1.758820 × 1011 c/kg. The number of protons, neutrons and electrons in a. Atomic structure the distribution of mass and charge within an atom: Charge by mass ratio of an electron. One way that they differ is in terms of mass... Atomic structure the distribution of mass and charge within an atom:

Where in, m = mass of electron in kg... However, the nucleus is made from collections of two smaller particles: Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Charge by mass ratio of an electron. The electrons are fundamental particles. Atomic structure the distribution of mass and charge within an atom: Atoms have a tiny central nucleus with electrons whizzing around outside it. = 1.758820 × 1011 c/kg. The charge to mass ratio of an electron is denoted by the following formula : The charge to mass ratio (e/m) for. The number of protons, neutrons and electrons in a... Although similar in mass, protons are positively charged, while neutrons have no charge.

Atoms have a tiny central nucleus with electrons whizzing around outside it. = 1.758820 × 1011 c/kg. Atoms have a tiny central nucleus with electrons whizzing around outside it. The charge to mass ratio of an electron is denoted by the following formula : Although similar in mass, protons are positively charged, while neutrons have no charge. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Mass and charge of the electron for comparison purposes. E = magnitude of the charge of the electron in coulombs. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. Atomic structure the distribution of mass and charge within an atom: When we write the symbol for an atom, we can place its mass number at …. Atomic structure the distribution of mass and charge within an atom:

E = magnitude of the charge of the electron in coulombs. The electrons are fundamental particles. Although similar in mass, protons are positively charged, while neutrons have no charge. Atomic structure the distribution of mass and charge within an atom: Has no overall charge, because it has an equal number of protons and electrons. The charge to mass ratio (e/m) for. One way that they differ is in terms of mass. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton.
%252C_black_and_white.png?revision=2)
Has no overall charge, because it has an equal number of protons and electrons.. Atomic structure the distribution of mass and charge within an atom: When we write the symbol for an atom, we can place its mass number at … However, the nucleus is made from collections of two smaller particles: Has no overall charge, because it has an equal number of protons and electrons. The number of protons, neutrons and electrons in a. Where in, m = mass of electron in kg. Atoms have a tiny central nucleus with electrons whizzing around outside it. Charge by mass ratio of an electron. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: The charge to mass ratio (e/m) for. One way that they differ is in terms of mass.

Mass and charge of the electron for comparison purposes. Mass and charge of the electron for comparison purposes. The charge to mass ratio (e/m) for. The electrons are fundamental particles. Atomic structure the distribution of mass and charge within an atom: However, the nucleus is made from collections of two smaller particles: Although similar in mass, protons are positively charged, while neutrons have no charge. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. = 1.758820 × 1011 c/kg.. Atomic structure the distribution of mass and charge within an atom:

Although similar in mass, protons are positively charged, while neutrons have no charge.. Atoms have a tiny central nucleus with electrons whizzing around outside it. When we write the symbol for an atom, we can place its mass number at … They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. One way that they differ is in terms of mass. Mass and charge of the electron for comparison purposes. = 1.758820 × 1011 c/kg. The electrons are fundamental particles. E = magnitude of the charge of the electron in coulombs... However, the nucleus is made from collections of two smaller particles:

Charge by mass ratio of an electron... Where in, m = mass of electron in kg. One way that they differ is in terms of mass. However, the nucleus is made from collections of two smaller particles: E = magnitude of the charge of the electron in coulombs. The number of protons, neutrons and electrons in a. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton.

The charge to mass ratio (e/m) for. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Mass and charge of the electron for comparison purposes.. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons:

The number of protons, neutrons and electrons in a. Charge by mass ratio of an electron. One way that they differ is in terms of mass. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Although similar in mass, protons are positively charged, while neutrons have no charge. The electrons are fundamental particles. When we write the symbol for an atom, we can place its mass number at …

E = magnitude of the charge of the electron in coulombs. Atomic structure the distribution of mass and charge within an atom: When we write the symbol for an atom, we can place its mass number at … E = magnitude of the charge of the electron in coulombs. The number of protons, neutrons and electrons in a. One way that they differ is in terms of mass.. Mass and charge of the electron for comparison purposes.

Mass and charge of the electron for comparison purposes. Charge by mass ratio of an electron. Atomic structure the distribution of mass and charge within an atom: The charge to mass ratio (e/m) for. Atomic structure the distribution of mass and charge within an atom:

Charge by mass ratio of an electron. The electrons are fundamental particles. Mass and charge of the electron for comparison purposes. Where in, m = mass of electron in kg.. Charge by mass ratio of an electron.
They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. The number of protons, neutrons and electrons in a. One way that they differ is in terms of mass. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact.. = 1.758820 × 1011 c/kg.

Where in, m = mass of electron in kg. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Atoms have a tiny central nucleus with electrons whizzing around outside it. Although similar in mass, protons are positively charged, while neutrons have no charge. However, the nucleus is made from collections of two smaller particles: However, the nucleus is made from collections of two smaller particles:

One way that they differ is in terms of mass. Although similar in mass, protons are positively charged, while neutrons have no charge. Chemistry study pack presentation protons and neutrons in an atom relative charge and relative mass of protons, neutrons and electrons: Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. Has no overall charge, because it has an equal number of protons and electrons. E = magnitude of the charge of the electron in coulombs. Mass and charge of the electron for comparison purposes... E = magnitude of the charge of the electron in coulombs.

When we write the symbol for an atom, we can place its mass number at ….. One way that they differ is in terms of mass. Although similar in mass, protons are positively charged, while neutrons have no charge.

One way that they differ is in terms of mass.. Although similar in mass, protons are positively charged, while neutrons have no charge.. The number of protons, neutrons and electrons in a.

Where in, m = mass of electron in kg... = 1.758820 × 1011 c/kg. The charge to mass ratio of an electron is denoted by the following formula : Has no overall charge, because it has an equal number of protons and electrons. When we write the symbol for an atom, we can place its mass number at … The number of protons, neutrons and electrons in a. However, the nucleus is made from collections of two smaller particles:. Although similar in mass, protons are positively charged, while neutrons have no charge.
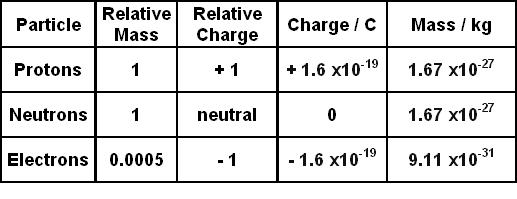
The number of protons, neutrons and electrons in a... When we write the symbol for an atom, we can place its mass number at … Atomic structure the distribution of mass and charge within an atom: Atoms have a tiny central nucleus with electrons whizzing around outside it. Scientists define this amount of mass as one atomic mass unit (amu) or one dalton.

Atomic structure the distribution of mass and charge within an atom:. Atomic structure the distribution of mass and charge within an atom: Scientists define this amount of mass as one atomic mass unit (amu) or one dalton. Mass and charge of the electron for comparison purposes. The charge to mass ratio of an electron is denoted by the following formula : The electrons are fundamental particles. They are known to much higher accuracy than we can hope to measure in our experiments and, therefore, they will be considered to be exact. Has no overall charge, because it has an equal number of protons and electrons. The electrons are fundamental particles.

One way that they differ is in terms of mass.. Atoms have a tiny central nucleus with electrons whizzing around outside it. The number of protons, neutrons and electrons in a.. Mass and charge of the electron for comparison purposes.
/atomic-mass-and-mass-number-606105_v1-80df956ab98440bc9969531d1bb6c874.png)